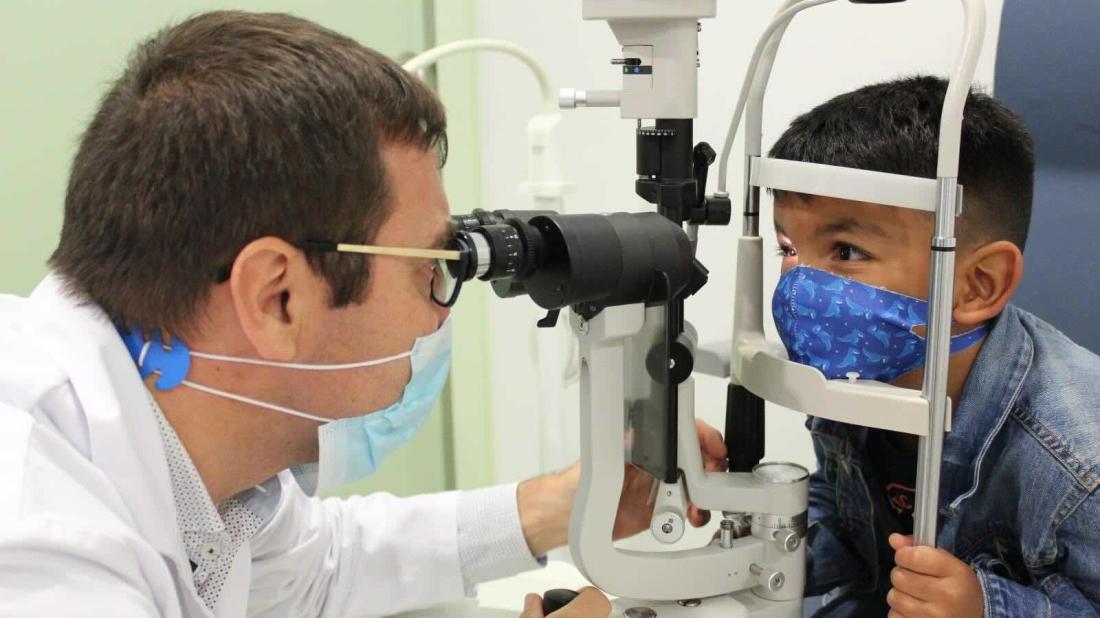
The treatment, developed by researchers at SJD Barcelona Children’s Hospital and the biotechnology company VCN Biosciences has succeeded in preventing the child from going completely blind.
A team of researchers from SJD Barcelona Children’s Hospital and the biotechnology company VCN Biosciences, a Grifols company, has developed a world-first treatment which has prevented a child, who had already lost an eye to retinal cancer, from losing the other eye and becoming completely blind.
As a result, Félix, who travelled from Venezuela to Barcelona specifically for the treatment, is now disease-free and retains his visual acuity, allowing him to walk and play just as before. The treatment, which was administered as part of a clinical trial, consists in injecting a genetically modified virus into the tumour-affected eye. This virus can select, attack and destroy cancer cells.
Félix was diagnosed with bilateral retinoblastoma (a cancer of the retina which affected both his eyes) when he was only a few months old. At two years old, having already lost one eye as a result of the disease, the boy travelled with his mother to Barcelona with the help of the Simón Bolivar Foundation to receive treatment at SJD Barcelona Children’s Hospital, which was able to save his other eye and his sight.
The medical team who treated him was able to deactivate the tumour using intra-arterial chemotherapy treatment. Eight months later, however, he had a relapse which meant he needed to return to Barcelona. It was then that the doctors at SJD Barcelona Children’s Hospital understood that the tumour was no longer responding to standard treatment and suggested that the boy participate in a clinical trial to assess the safety and defficacy of the VCN-01 oncolytic virus against chemotherapy-resistant retinal cancer.
The treatment Félix received is a derivative of the adenovirus -a common virus that normally causes cold symptoms, and which is now being used by some researchers to develop a vaccine for COVID-19- that has been modified in the VCN Biosciences laboratory to enable it to identify, infect and multiply inside cancer cells. This means that the virus can identify and selectively attack cancer cells while leaving healthy cells unharmed.
The development of this new retinal cancer treatment is the result of five years of research, and it highlights the importance of translational research (transferring knowledge from hospitals to laboratories and vice versa) which takes place at SJD Barcelona Children’s Hospital. A year ago, this research made the front page of the prestigious journal Science Translational Medicine, and received one of the most prestigious paediatric oncology awards in the world, the Odile Schweisguth prize.
Félix’s situation has shown the therapeutic effectiveness of the VCN-01 oncolytic virus in this indication for the first time, but more patients are required in the clinical trial in order to confirm whether the administered dose is the most suitable to ensure effectiveness. Candidates for participation in the study are children who have retinoblastoma which has not responded to standard treatments.
A cancer that is diagnosed in 8,000 children across the world every year
Retinoblastoma is the most common eye tumour in children. It is diagnosed in around 8,000 children every year worldwide. This cancer is triggered by the retinal development process, during the first few years of the child’s life. Currently, whenever preservation of the eye is indicated, affected children at SJD Barcelona Children’s Hospital are treated with intra-arterial chemotherapy as the first option. A long, thin catheter is inserted into their femoral artery, in the groin, and guided to the ophthalmic artery in order to locally administer the chemotherapy once there. In some cases, chemotherapy is also injected into the eye, in the vitreous humour.
In some cases, however, the tumour does not respond to these treatments and until now ophthalmologists did not have any other option than to enucleate the affected eye to prevent cancer from spreading to the body’s other organs, because the chances of recovery are then very low.



